Research Topic
2. Redox regulation of electron flow-Ions and photosynthesis
Giovanni Finazzi
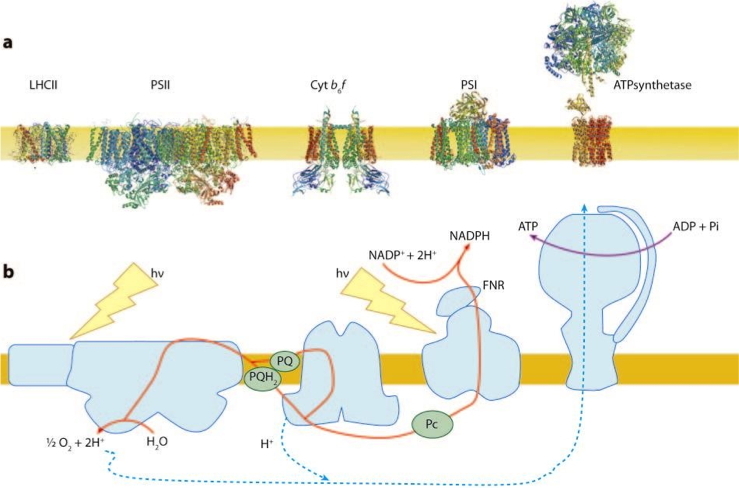
Most biochemical reactions in living cells are fuelled by adenosine triphosphate (ATP) and/or the reducing agent reduced nicotinamide adenine dinucleotide phosphate (NADPH). In plant cells, these components are generated in mitochondria and chloroplasts through the respiratory and photosynthetic electron transfer chains. Nearly 50 years ago, Mitchell proposed a unifying mechanism through which the respiratory and photosynthetic electron transfer reactions are coupled to proton pumping across the inner mitochondrial and thylakoid membranes; the thylakoid membranes are located within chloroplasts and contain the protein-pigment complexes that catalyze the primary reactions of photosynthesis (Figure 3).
Figure 3:
a - Transmembrane organisation of the major photosynthetic proteins in their native oligomerization state.
b - Schematic representation of the pathway for photosynthetic electron flow.
This proton movement generates a transmembrane electrochemical gradient called the proton motive force (pmf), which is comprised by a proton gradient (ΔpH) and an electric field (ΔΨ). While both components of the pmf are equally necessary for ATP synthesis, the ΔpH specifically regulates photosynthetic control and non-photochemical quenching (NPQ, Figure 1) by modulating both the protonation state of key regulatory proteins and the rate of electron flow in the cytochrome b6f complex. It is therefore assumed that the size of the ΔpH must be regulated in response to environmental stimuli through a still elusive mechanism. Our previous study suggests that potassium flux across the thylakoid membrane through an ion channel (TPK3 Checchetto et al., 2012, Carraretto et al., 2013) regulates the proton motive force by counterbalancing the H+ gradient with K+ flux and thus impacts photoprotection by modulating non photochemical quenching (NPQ) and electron flow, ultimately affecting plant fitness
QUESTIONS
Is it possible to optimize photosynthesis fluxes in algae (Chlamydomonas reinhardtii), mosses (Physcomitrella patens, Marchantia polymorpha) and plants (Arabidopsis thaliana) by regulating ion fluxes? To answer this question, we are using a comprehensive and integrative approach, to identify new players and dissect the coordinate regulation of light conversion into chemical energy. To this aim we are combining our expertise with competences of our collaborators (electrophysiology, bioinorganic chemistry and molecular genetics) to identify putative chloroplast ion channels, generate down-regulated mutants, characterize their function in vitro (electrophysiology) and in vivo (using a home-made biophysical tool: Stark spectroscopy) and collate their phenotype to ion dynamics in vivo (by development and application of new fluorescent sensors for imaging metal ion fluxes in living plant cells and whole organisms). This research is funded by a Human Frontier Science Program (HFSP), which started in September 2015 and gathers a consortium of laboratories from Japan (University of Kyoto), USA (University of Berkeley) and Europe (University of Padova, Italy and our team).